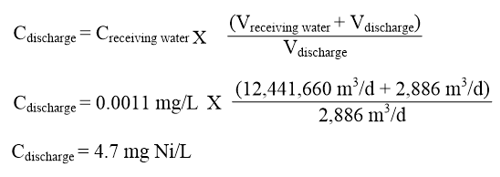2.1 WHY ACCOUNT FOR BIOAVAILABILITY?
The Priority Substances Daughter Directive states that when assessing monitoring results against the EQS for metals account may be taken of:
- natural background concentrations for metals and their compounds where such concentrations prevent compliance with the relevant EQS; and
- hardness, pH, dissolved organic carbon, or other water quality parameters that affect the bioavailability of metals.
Accounting for bioavailability represents the most technically robust method for assessing the potential risk of nickel in the freshwater aquatic environment. Bioavailability is a function of the various physicochemical factors governing metal availability at the biological receptor (Drexler et al. 2003). Effectively this means that bioavailability reflects the actual metal exposure to an aquatic organism. For many metals, including nickel, adverse effects on aquatic organisms in the water column are assumed to be as a consequence of exposure to the bioavailable Ni2+-ion. This is important as it has long been established that total (dissolved and undissolved) metal concentrations in waters have limited relevance to potential environmental risk (Campbell 1995; Niyogi and Wood 2004). In addition, hardness-based EQS for metals are also poor metrics of ecological risk (The Netherlands 2004).
The misidentification of potential risks in surface waters can result in inappropriate regulatory intervention (Environment Agency 2009). Subsequent regulatory actions (i.e., Programs of Measures under the WFD) based on existing EQS have the potential to result in considerable financial implications that are likely to deliver limited ecological benefit.
The conclusions of the EU RAR were scientifically underpinned by the development of chronic biotic ligand models (BLM) (see Fact Sheet 4 for additional information). These models, by taking account of site-specific water chemistry, relate toxicity to bioavailable nickel concentrations, rather than total nickel or dissolved nickel concentrations. The use of BLMs for deriving and implementing metal EQS was supported by the Scientific Committee on Health and Environmental Risks (SCHER) in their comments on the EQS Technical Guidance (European Commission 2010), although they acknowledge that accounting for bioavailability in a regulatory compliance framework would mean a step-change in the regulatory paradigm.
The revision of the nickel EQS represents the first time that the development and implementation of a bioavailable EQS using chronic BLMs has been undertaken in a regulatory framework at a European level.
2.2 PREDICTED NO EFFECT CONCENTRATION VERSUS EQS?
Predicted No Effect Concentrations (PNECs) for freshwater have been calculated in the EU RAR under several bioavailability scenarios. These scenarios are described in greater detail in Fact Sheet 4 but effectively they represent water conditions at seven locations within the EU that reflect a range spanning the 10th to 90th percentile of the physicochemical water chemistry conditions across the EU. However, the PNECs used in the EU RAR are different from a final nickel EQS that will be derived for the WFD. The key differences are that a PNEC is not legally binding and is part of an iterative risk assessment process, meaning that refinement is possible if risks are identified. In addition, an overly conservative PNEC is of little concern if no risks are identified. Alternatively, an EQS under the WFD is:
- legally binding,
- must be protective in all situations (defined in the EQS Technical Guidance as representative of at least 95 percent of the surface waters), and
- not overly stringent as there is no capability to refine the value if it is exceeded.
In addition, the EU Commission and Member States prefer a single EQS value for a substance to cover the whole of Europe, i.e., a generic EQS not multiple EQS across Member States. This preference limits the possibility of setting site-specific EQS to account for differences in bioavailability, and instead means that correction for bioavailability should be made by normalizing exposure concentrations to “bioavailable metal.”
Therefore there is a need to derive a generic nickel EQS that builds on the understanding of bioavailability made in the EU RAR, but that in principle will be protective for all waterbodies monitored in the EU.
2.3 DERIVING A NICKEL EQS UNDER THE WFD
In addition to a numerical value, a robust EQS must also meet other implementation requirements (Crane et al. 2010) to ensure that it can be used within a regulatory framework. These requirements may include considerations of analytical sensitivity or uncertainty, the need to collect data on supporting parameters (e.g., those required to support bioavailability calculations) or any requirements for data processing (i.e., undertaking bioavailability calculations).
The EU EQS Technical Guidance advocates the use of a tiered approach (e.g., Figure 1). A tiered approach allows low priority sites to be removed from the assessment at early tiers, whilst concentrating regulatory (and financial) effort on higher priority sites. The first tier in this approach does not explicitly consider bioavailability and uses a generic EQS to which annual average dissolved nickel concentrations are compared. If a site fails a compliance assessment based on the generic EQS then it is subject to further evaluation before any decision on compliance is made.
The EC recommends that the generic EQS is set to reflect the conditions of maximum bioavailability which are likely to be observed for the region of application (European Commission 2010). For nickel, high bioavailability conditions are generally observed in waters with relatively high pH, low hardness, and low dissolved organic carbon (DOC).
In order to determine the generic EQS for nickel in EU waters it is necessary to identify the conditions that reflect maximum bioavailability in the EU, i.e., those conditions that result in the lowest HC5 values.1 Based on the EU RAR, the scenario that had the greatest bioavailability was that for Lake Monate in Italy. The HC5 for this waterbody was 7.2 µg/L (based on a pH of 7.7, 48 mg Ca/L and 2.5 mg DOC/L). Table 1 reports nickel PNECs for additional European freshwater scenarios based on regional datasets for Great Britain, France, Austria, Spain, The Elbe, Sweden, and Walloon (Belgium).
It is important to note that the PNECs reported in Table 1 are the HC5 values with an Assessment Factor (AF) of 1. This is in line with the nickel EQS as proposed under the amended Daughter Directive (2013/39/EU). This is a change from the EU RAR where the absence of field and/or mesocosm data led the Technical Committee for New and Existing Substances (TCNES) to propose that an AF of 2 should be used in the EU RAR PNEC derivation process to account for residual uncertainties in the assessment. However, since the completion of the EU RAR, mesocosm (Hommen et al. 2011) and field data (Peters et al. 2013) have been generated and this has been judged by Member State Experts and DG Environment to have reduced uncertainty to a level whereby an AF of 1 is acceptable.
The lowest of the 5th percentiles of frequency distributions of site-specific PNECs reported in Table 1 are approximately 4 µg/L (at pH 8.2, DOC 2 mg/L, Ca 40 mg/L). Therefore, setting the generic EQS as 4 µg/L as finalized in the amended Daughter Directive ensures that the first tier of the compliance assessment is adequately protective of most conditions likely to be encountered in the EU.
The generic nickel EQS is expressed as “bioavailable nickel” (i.e., generic EQSbioavailable). The use of the term generic EQSbioavailable is effectively the same as defining an EQS on the basis of a specific form (or species) of a chemical. A generic EQSbioavailable can only be used in a tiered approach in which bioavailability is accounted for in at least some of the tiers. Using the generic nickel EQSbioavailable as a single pass/fail criterion without accounting for bioavailability (i.e., assuming that all measured dissolved metal is bioavailable) would result in many sites erroneously being identified as being at risk (Figure 1).
1 As discussed in Fact Sheet 1, the PNEC is calculated as PNEC = HC5/AF, where:
HC5 = hazard concentration at the 5th percentile of the Special Sensitivity Distribution and
AF = assessment factor
The EQS is equivalent to the HC5
2 The 2010 Draft Nickel EQS Dossiers are available on request from NiPERA. Please contact Emily Garman at egarman@nipera.org.