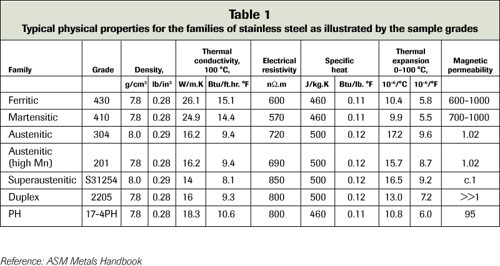
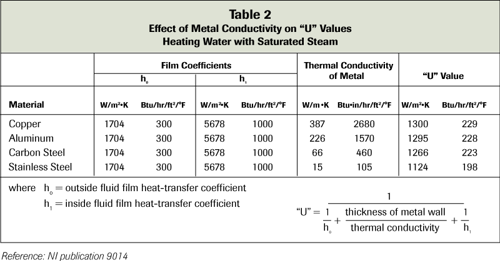
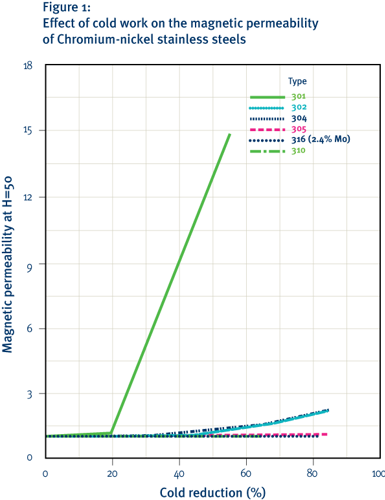
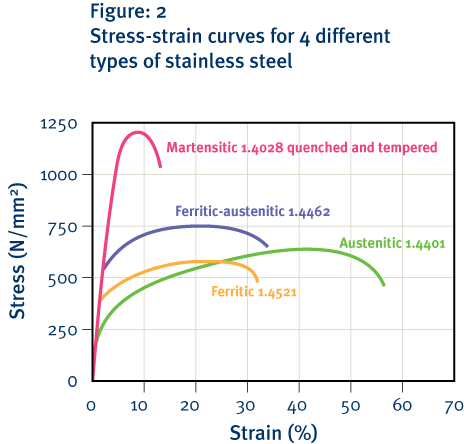
-
Formability:
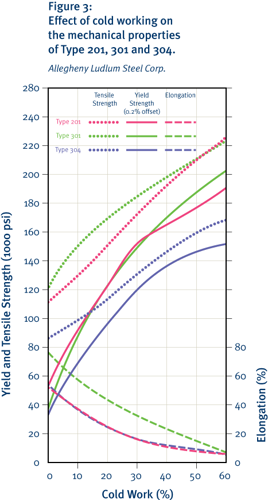
The characteristics of the austenitic structure give these stainless steels good tensile ductility and good formability, as reflected in comparative forming limit diagrams. The common 18 percent chromium/8 percent nickel grade shows particularly good stretch-forming characteristics but with a somewhat lower limiting drawing ratio than some ferritic grades. Slightly higher nickel contents increase the stability of the austenite further and reduce the work-hardening tendency, thereby increasing suitability to deep drawing. Unlike traditional low-nickel, high-manganese grades, these are not prone to delayed cold cracking. This good formability has led to the widespread use of 300-series austenitic grades for items such as kitchen sinks, pots and pans.
-
Weldability:
Many pieces of equipment have to be fabricated by welding. In general, the nickel austenitic grades have superior weldability to other grades, and Types 304 and 316 are the most widely-fabricated stainless steels in the world. They are not prone to becoming brittle as a result of high-temperature grain growth and the welds have good bend and impact properties. They are also more weldable in thick sections of, say, above 2 mm.
The duplex grades are far more weldable than the ferritics for equivalent alloy content, but even the standard and more highly-alloyed super-duplex alloys require greater attention to the details of the welding procedure than the equivalent austenitic grades. The 200-series alloys have welding characteristics similar to the 300 series.
-
Toughness:
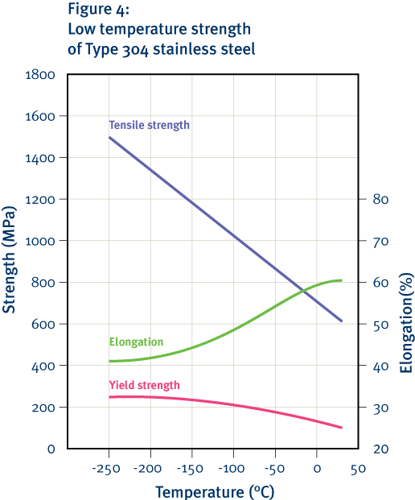
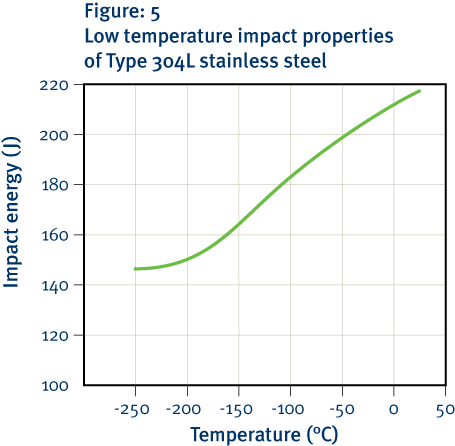
The ability of a material to absorb energy without breaking is essential in many engineering applications. Most stainless steels have good toughness at room temperature; however with decreasing temperature, the ferritic structure becomes progressively more brittle, making ferritic stainless steels unsuitable for use at cryogenic temperatures. In contrast, the common austenitic stainless steels retain good toughness even at liquid helium temperatures; therefore grades such as Type 304 are widely used for cryogenic applications.
-
High-Temperature Properties:
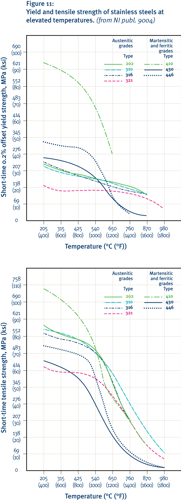
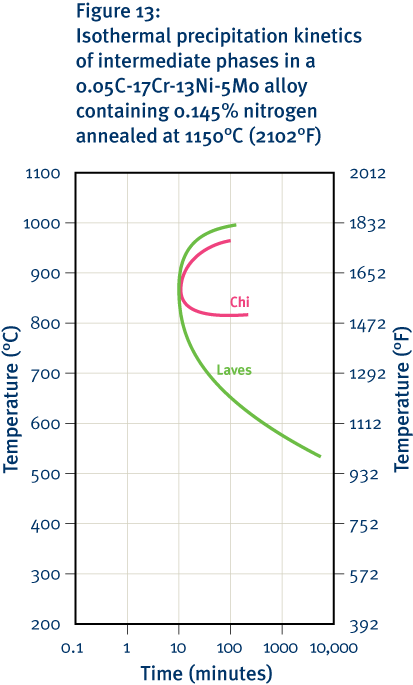
The addition of nickel gives the austenitic grades significantly better high-temperature strength than other grades, particularly the ability to resist creep. These grades are also much less prone to the formation of deleterious phases as a result of exposure at intermediate and high temperatures. Nickel also promotes the stability of the protective oxide film and reduces spalling during thermal cycling. Consequently, the austenitic grades are favoured for high-temperature applications and where fire resistance is needed.
It is worth noting that there is a continuum in composition between the austenitic stainless steels and the nickel-based superalloys used for the most demanding high-temperature applications, such as gas turbines.
-
Corrosion Resistance:
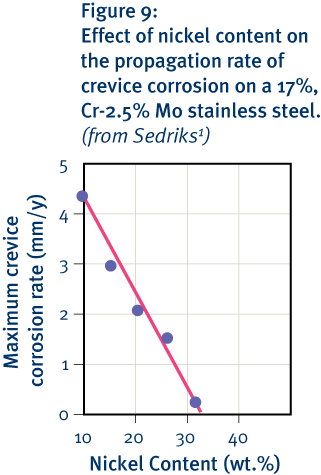
As noted, it is the chromium-rich oxide layer that chiefly accounts for the corrosion resistance of stainless steels. However, this layer is susceptible to damage, particularly in the presence of chlorides, and such damage can lead to the onset of localised corrosion such as pitting and crevice corrosion. Both molybdenum and nitrogen increase resistance to pit initiation in the presence of chlorides. Nickel does not influence the initiation phase but is important in reducing the rate at which both pitting and crevice corrosion propagate (see Figure 9). This is critical in determining how serious corrosion will be.
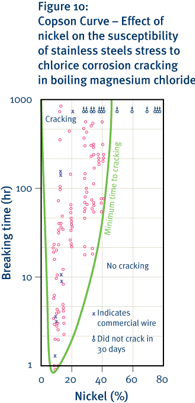
Nickel also influences the resistance of stainless steels to another form of localised corrosion, namely chloride stress-corrosion cracking. In such cases, however, there is a minimum resistance at nickel contents of around 8 percent. Stress corrosion-cracking resistance increases markedly at nickel levels both lower and higher than this.
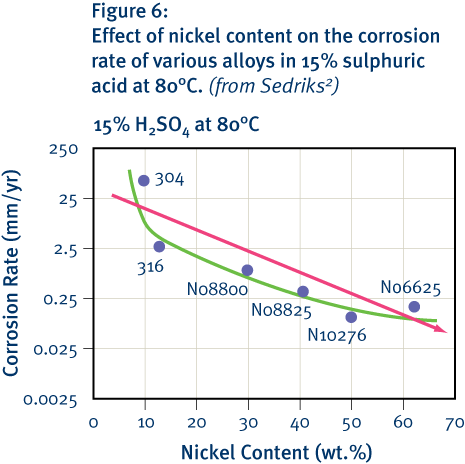
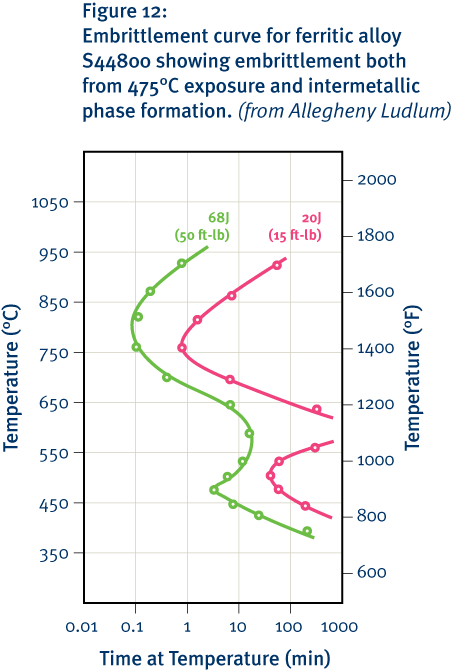
In general, increasing the nickel content of stainless steels, including ferritic grades, also increases their resistance to reducing acids such as sulphuric acid. Other elements, including molybdenum and particularly copper, also have a strong influence in this regard. However, there are potential drawbacks to using nickel in this way in the ferritic grades. These drawbacks relate to stress corrosion-cracking resistance and the formation of intermetallic phases.
-
Lustre and Finish:
At first sight, all stainless steel grades look similar. However, side-by-side comparisons of identically polished surface finishes do show differences in colour and lustre. Appearance and aesthetic qualities will always be a matter of taste; the 200-series grades generally appear darker and the ferritic grades cooler-looking, than the nickel austenitic grades. In some architectural applications, a greyer colour might be preferred, but consumers generally prefer a brighter, whiter metal, as witnessed by the popularity of the 300-series. The 200-and 300-series stainless steels are also more scratch-resistant, owing to their inherent work hardening properties.
Various surface finishes are available on all stainless grades, from mill finishes to mechanically polished (rough to mirror-finished), brushed, bead-blasted, patterned and many more. This emphasises indicates the versatility of the nickel stainless steels in achieving a wide range of aesthetic appearances. One caveat, however, is that a rougher finish will generally have poorer corrosion resistance, particularly in outdoor architectural applications. Marine environments and the presence of de-icing salts require more corrosion-resistant materials, such as Type 316L.
-
Sustainability:
Taking into account the Brundtland Report’s definition of sustainable development - “development that meets the needs of the present without compromising the ability of future generations to meet their own needs” - it is clear that stainless steels in general, and the nickel-containing ones in particular, can play a major role in the areas of environmental protection, economic growth, and social equality. Some examples are given below.
To appreciate the contribution a material makes toward sustainability, it is important to examine that material’s whole life cycle, from extraction to recycling or disposal at the end of the product’s life.
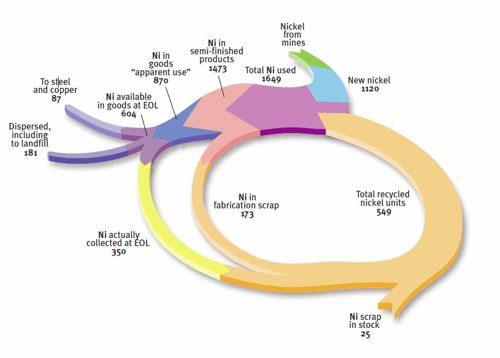
Most nickel-containing materials are fully recyclable at the end of a product’s useful life; in fact the high value of nickel encourages this. Recycling lessens the environmental impact by reducing both the need for virgin raw materials and the use of energy for production. For example, the amount of stainless steel scrap being used today reduces the energy required for the manufacture of stainless steel to about one-third less than would be required if 100 percent virgin materials were to be used (Yale ). Nearly half of that reduction comes from end-of-life scrap (using IISF data ). Only a lack of availability of more scrap, owing to the extended useful life and considerable growth in the use of stainless steel products, prevents a greater reduction.
The key contributions of nickel-containing stainless steels are that, when properly applied, they maintain and improve the quality of life of citizens and allow businesses and other institutions to deliver sustainable solutions. These sustainable solutions rely on the attributes and services provided by nickel: corrosion protection, durability, ease of cleaning, temperature resistance and recyclability.
The most visible examples of the durability of stainless steels are in buildings. The restorations of St Paul’s Cathedral and the Savoy Hotel canopy in London, U.K. (1925 and 1929, respectively), the Chrysler Building in New York City (1930), the Progreso Pier in Mexico’s Yucatan state (c. 1940), the Thyssen Building in Düsseldorf, Germany (1960) and the Gateway Arch in St Louis, U.S.A. (1965) are all testament to the long life delivered by nickel-containing stainless steel.
-
Ease of production:
This aspect is not something that is immediately apparent to the final user. However, the long experience of manufacturing the common austenitic grades, their widespread use, their versatility and the scale of their production mean that they have become commodity grades of a high quality. These grades are economically available in all forms and in all parts of the world.
-
Stainless steel in use:
The picture that emerges is of the common nickel-containing austenitic grades being good all-round performers. They are widely available, well-understood, versatile and easy to use. They also demonstrate high performance and are extensively recyclable. All of which means they often provide the most practical, lowest-risk solution.
As they have been in use for so long, the 300 series grades are often already approved for use in situations that involve contact with food or drinking water. In addition, all product forms needed are usually readily available.